The coming will always come. Sun Wen is only one thing for one thing - revolution. The revolution is not utopia, but the opposite is full of "copper smell." In 1894, he ran to Honolulu to organize the Xingzhong Association. In essence, he was an institution that raised money. This time, it raised a sum of 13,000 Hong Kong dollars, so he ran to Guangzhou to organize the first uprising. The war spent money like water, so Sun Wen kept going overseas. Of course, in order to raise money, he used various means. If you look for your own big brother, Sun Mei, this is a farmer and animal husbandry who made a family, and people called it "Maui Island King". In 1895, the rich man sold his livestock for his brother-in-law to donate his army. Later, "for money." In 1909, the "China Red Dragon Program" agreed that $3.5 million would give the uprising organization authority to Charlie Booth, and that the league would pay an annual interest rate of 15%, pay off the principal and interest within ten years, and vacate the US consortium businessman in the new government. Some important positions were later aborted. Another example is the issuance of stocks and bonds. In May 1911, the Confederation's Chicago branch rally announced the establishment of a "revolutionary company" to mobilize the purchase of the "company" stock, and doubled the reimbursement after the success of the revolution; however, Chicago had little effect, Sun Wenyi transferred to Vancouver, and issued another 10 yuan. , 100 yuan, 1000 yuan of gold coins bonds for support. Fortunately, the coming is finally coming, a large number of "crowdfunding" finally returned to the intermittent success of the revolution - Wuchang Shouyi. Sun Wen became the interim president, and the arrears account was reimbursed to the revolutionary government. The blood of the martyrs has injected vitality into China, and the current new medical policy has also injected an upgrade into the industry. For example, in 2017, the “Opinions on Deepening the Reform of the Examination and Approval System and Encouraging the Innovation of Drug Medical Devicesâ€, No. 42, No. 44, etc., the review and approval speed-up documents, the industry has almost entered the “national research and developmentâ€, from time to time, new drugs, clinical, Listing... Even the HKEx is “lowering†the threshold for unprofitable biotech companies. Under this circumstance, the valuations of pharmaceutical companies in A and H have also skyrocketed, and they have been pulled at least 10 times in the past year alone. However, because of the defensive role, it seems that "expensive" has to bite his teeth and insist on killing. After all, the market for drinking and taking medicine has no stop. The market continues to improve, and the A and H medical sectors are insane. Zhitong Finance APP observed that as of May 28, the Hong Kong stock health care index continued to rise, up 1.19%. Among them, Sansheng Pharmaceutical (01530) led the constituent stocks, which rose more than 6 points, and the market value increased sharply by 33 billion Hong Kong dollars a day. Source: Futu Securities Acquired good products to create a new history When it comes to pharmaceutical companies, there are only two ways: one is to work hard to do research and development; the other is to merge and “secondaryâ€. Investors who are familiar with Sansheng Pharmaceuticals are very clear that they are biopharmaceutical companies that have come out to do research and development. Because from the financial data side, in 2012-2017, the company's operating income, gross profit and net profit margin achieved a compound annual growth rate of 41.6%, 39.1% and 57.7% under the dual role of its own products and acquired varieties. In particular, the gross profit margin of core products has remained above 90%, and the market share has continued to increase. In view of the good results of the acquisition, in 2016, Sansheng Pharmaceutical acquired a 100% interest in Zhejiang Wanhao, which provides dermatological drugs, anticancer drugs and drugs for the treatment of diabetic complications, with a revenue of 103 million yuan. Then, in March, based on the equity of CITIC Guojian 54.36, Sansheng Pharmaceutical increased its holding of CITIC Guojian to 97.78% with 3.932 billion yuan and renamed it "Sansheng Guojian". Spend 3.932 billion yuan to "strongly eat" CITIC Guojian, Zhitong Finance APP has repeatedly analyzed, is to get a few products in the hands of CITIC, because this target in addition to possession of Yisaipu (etasicept) and Jian Nipera In addition to the two listed products, there are three 750-liter and two 3000-liter reactors. If all the grafts are smooth, the economic benefits will not be lost. In June, Sansheng Pharmaceutical invested US$10 million to establish a CAR-T therapy joint venture with US biotechnology company Sorrento Therapeutics and will acquire 51% of its shares. The joint venture will acquire exclusive licenses for three immunotherapeutic technologies from Sorkento's subsidiary TNK in mainland China, Hong Kong and Macau. On October 11, Sansheng Pharmaceutical affiliated Hong Kong Sansheng entered into an exclusive license agreement with AstraZeneca. Pursuant to the agreement, AstraZeneca agreed to grant Hong Kong Sansheng exclusive rights to commercialize licensed products (four types of diabetes control products: Byetta, Bydureonsingledosetray, Bydureondualchamberpen, Bydureonautoinjector), and as a consideration for this exclusive right, Hong Kong Sansheng agreed AstraZeneca pays a prepayment of $50 million and a maximum installment of $50 million. This is definitely a heavy product for Sansheng Pharmaceutical. Zhitong Finance APP learned that IDF expects that there are currently about 114 million adults with diabetes in China, most of which are type 2 diabetes, and about 53.6% of people with diabetes have not been accurately diagnosed. From this data, by 2025, the diagnosis rate of Chinese diabetic patients can be increased to 60%, and the treatment rate has increased from 30% in 18 years to 40%. To date, due to the high cost of treatment, the penetration rate of GLP-1 receptor agonists is less than 1%. If covered under medical insurance (the “Guidelines for the Prevention and Treatment of Type 2 Diabetes in China (2017 Edition)†released in November 2017 will be GLP- 1 Receptor agonist is recommended as a second-line treatment for type 2 diabetes.), the penetration rate of GLP-1 receptor agonist will reach 10% in 2025. In other words, the peak sales of Baidayang are expected to exceed 880 million yuan. As predicted, Sansheng Pharmaceutical has become crazy after its official listing. As of May 28, it opened higher and higher, rose 6.3% throughout the day, reported 21.95 Hong Kong dollars, the stock hit a new high, the net inflow of more than 65.38 million Hong Kong dollars, is definitely the most brilliant day in the history of Sansheng Pharmaceutical. Source: Futu Securities Herceptin or the future breeze In fact, during the acquisition process, Sansheng Pharmaceutical did not give up its own research and development. Zhitong Finance APP observed that as of now, the company has 31 products under research, including 16 countries and 1 class of new drugs. 6 kidney products, 7 oncology products, 11 autoimmune and other disease products, 3 products for metabolic diseases such as diabetes; 4 dermatology products. Among them, leading bio-products at different stages of clinical development, including NuPIAO (second-generation rhEPO products for the treatment of anemia), SSS07 (anti-tumor necrosis factor alpha antibody products for the treatment of rheumatoid arthritis), Pegsiticase (a An improved polyethylene glycol recombinant uricase derived from Candida utilis for the treatment of refractory gout), 602 (an anti-epidermal growth factor receptor antibody for the treatment of cancer), 601 For the treatment of AMD anti-VEGF antibody) and Yisaipu pre-filled injection. Herceptin is actually a similar drug developed by Roche for the treatment of HER2 overexpressing breast cancer, metastatic gastric cancer or esophagogastric junctional adenocarcinoma. About Herceptin, the United States also shot a film called LivingProof, which tells the story of Slammon's development of the drug. On September 25, 1998, Roche's Herceptin (trastuzumab) was approved by the FDA and entered the Chinese market on September 5, 2002. As of 2014, its European patents expire and US patents will expire in June 2019. Although the patent is about to expire, Roche relies on the drug to make nearly $10 billion a year. For example, in 2016, the drug achieved revenue of 6.782 billion Swiss francs (about US$7.034 billion), a 4% increase year-on-year, and ranked 8th in global sales. Let me talk about SSS07 humanized anti-TNFα antibody. In the second quarter of 2017, the product has started the Phase Ib clinical trial in patients with rheumatoid arthritis. Preclinical data show that the efficacy and specificity of SSS07 is better than the existing similar products. At present, the number of rheumatoid arthritis patients in China is about 4.5 million. It is expected that the number will continue to rise with the aging of the population, while the penetration rate of anti-TNF drugs in the field of rheumatoid arthritis is less than 1%. In 2016, the market size of anti-TNF drugs was about 1.74 billion yuan. Benefiting from the coverage of new medical insurance in 2017, the affordability of drugs has been greatly improved. It is estimated that by 2020, the market size will increase to 4.29 billion yuan, and the CAGR will be 25.3% in 16-20 years. Simply understand, Sansheng Pharmaceutical's product is also over 100 million varieties. In the end, in the short term, the original products, especially Patek, are enough to maintain the growth of Sansheng Pharmaceutical's “firepowerâ€; in the long run, heavy research products will strengthen its moat. Both long and short-term, it indicates that the company has great potential. After all, it will always come. (Tian Yuxuan / Wen) This article was first published on WeChat public account: Hong Kong stock excavator. The content of the article belongs to the author's personal opinion and does not represent the position of Hexun.com. Investors should act accordingly, at their own risk. (Editor: Yue Right HN152) Diabetic Foot Care Socks,Diabetic Socks,Diabetic Compression Socks,Diabetic Ankle Socks Hengshui Yiqingshu Hosiery Co., LTD. , https://www.yiqingshusocks.com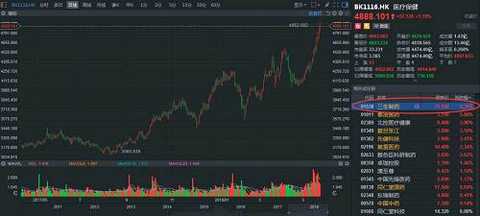
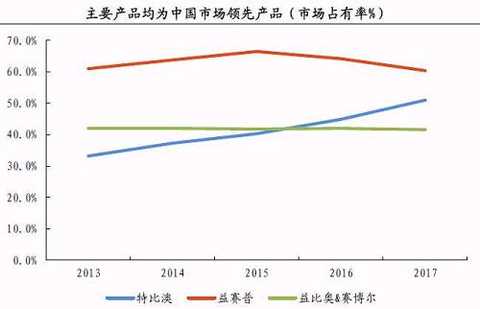
Before returning to the market, in November 2014, Sansheng Pharmaceutical obtained the development rights of the oncology drug tanibirumab in China from Pharm Abcine, Korea. In December of the same year, the company acquired 100% of the rights of Guangdong Saibaoer. Saibaoer mainly provides recombinant human erythropoietin and low molecular weight heparin injection. In 2015, it will bring 39 million yuan to Sansheng Pharmaceutical. 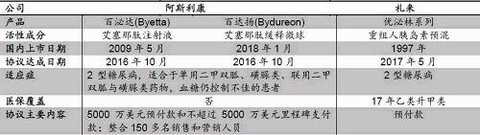
Among them, the first glucagon-like peptide-1 (GLP-1) receptor agonist weekly preparation Baidayang (common name: exenatide microsphere for injection) was officially acquired on January 4, 2018. CFDA approved and officially listed in China on May 25, the first weekly hypoglycemic agent in China. 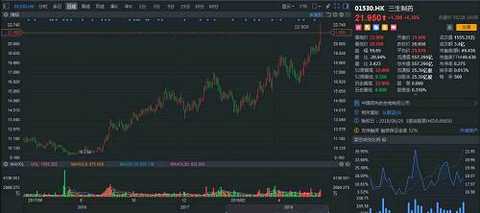

However, the well-received Xaptin and Jiantuo were previously retired due to clinical self-examination, but because these two are heavy products, Sansheng Pharmaceutical is also a re-submitted heavy new drug application - trastuzumab ( Herceptin's application, based on time projections, is expected to be available in the mid to late 2018. 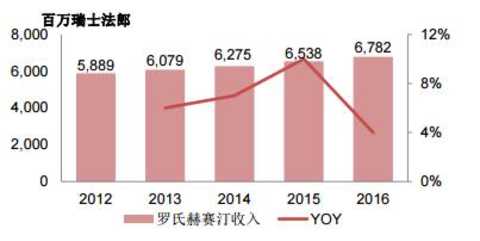
A variety can bring tens of billions of dollars in revenue. If the 20-year-old Trioxon's trastuzumab can be listed, it is undoubtedly expected to become the earliest listed HER2 product in China, which is definitely a big profit.